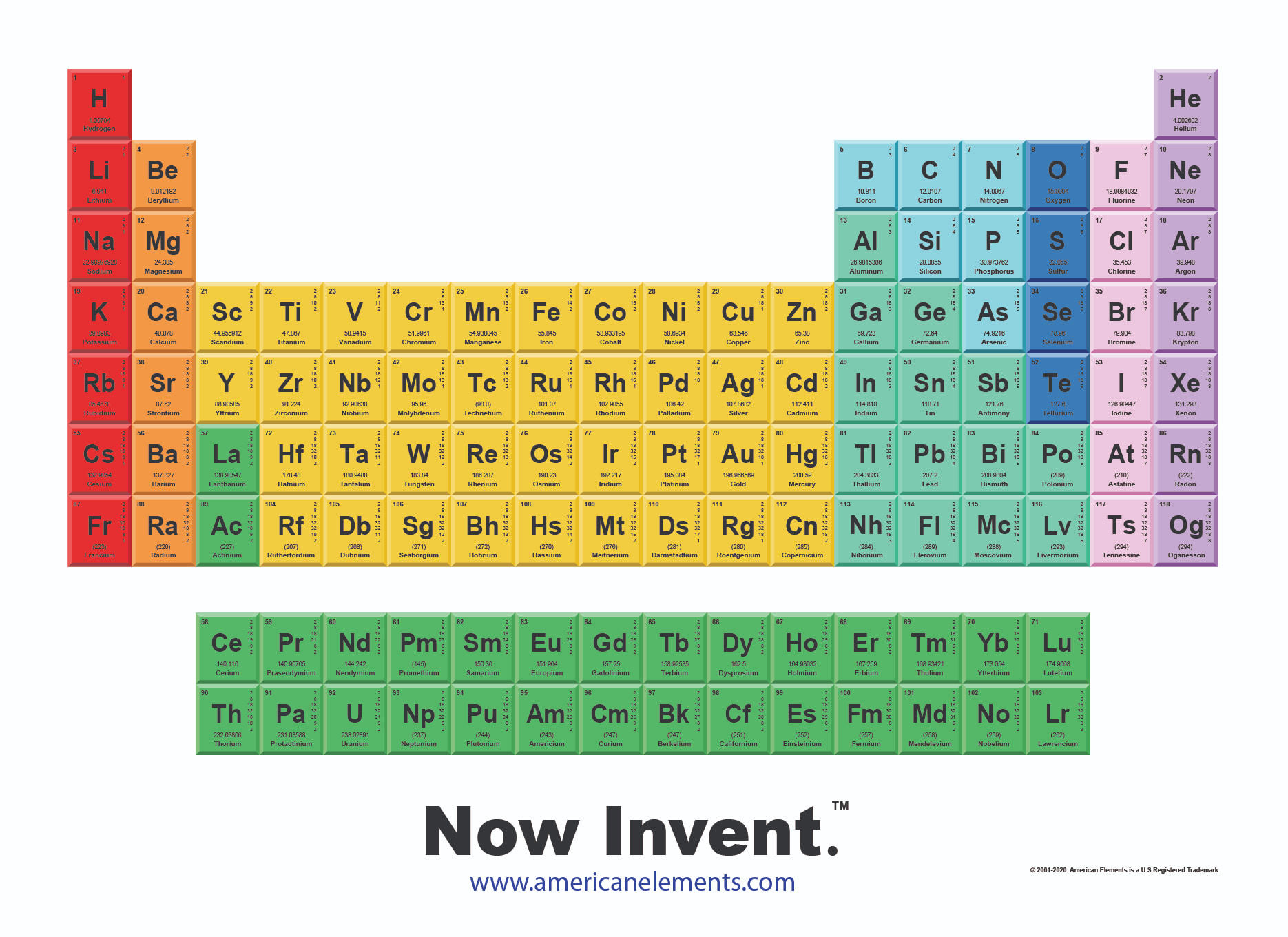
When an organism dies, it stops taking in carbon-14, so the ratio of carbon-14 to carbon-12 in its remains, such as fossilized bones, will decline as carbon-14 decays gradually to nitrogen-14 2 ^2 2 squared. As animals eat the plants, or eat other animals that ate plants, the concentrations of carbon-14 in their bodies will also match the atmospheric concentration. The modern periodic table is based closely on the ideas he used: the elements are shown by the symbol for their atom. In a periodic table arranged in order of increasing atomic number, elements having similar chemical properties naturally line up in the same column (group). As plants pull carbon dioxide from the air to make sugars, the relative amount of carbon-14 in their tissues will be equal to the concentration of carbon-14 in the atmosphere. These forms of carbon are found in the atmosphere in relatively constant proportions, with carbon-12 as the major form at about 99%, carbon-13 as a minor form at about 1%, and carbon-14 present only in tiny amounts 1 ^1 1 start superscript, 1, end superscript. Now available: history of the periodic table. The lanthanide series includes elements with atomic number 57 to 71. The last two periods are somewhat unique, because they contain the lanthanide and actinide series. The second and third periods contain 8 elements each, and the fourth and fifth periods contain 18 elements each. There is also a 2019 edition of this table. Here is the pdf file of the color periodic table so you can save and print it. It includes element names, symbols, atomic numbers, atomic weights, element groups, and periods. It has a unique arrangement of rows and columns. 2013 Edition This free periodic table wallpaper has a white background. The periodic table, electron shells, and orbitals. Each chemical element contains a link to a page that explains its chemical properties, health effects, environmental effects, application data, an image and also information of the history/inventor of each element. The first period only contains two, hydrogen (H) and helium (He). The periodic table is a way of organising the elements which is used by scientists to group elements with similar properties. Atomic structure and electron configuration. For example, carbon is normally present in the atmosphere in the form of gases like carbon dioxide, and it exists in three isotopic forms: carbon-12 and carbon-13, which are stable, and carbon-14, which is radioactive. Periodic table - chart of all chemical elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
